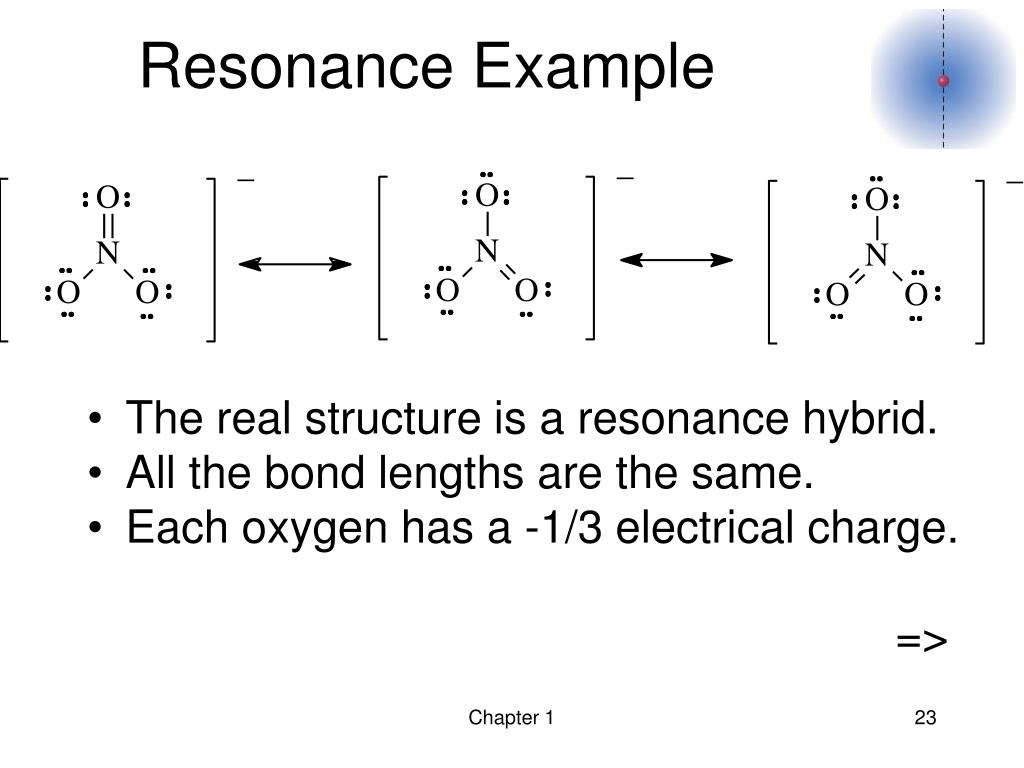 
Resonance hybrids must have the same number of unpaired electrons (if exists). This extended model led to the concept of resonance, as the coupling between Lewis structures stabilizes the hybrid structure, in analogy to Heisenbergs model of the helium atom resembling a. In simple words resonance energy is the energy that helps in determining the delocalized electrons present in any molecules where a single Lewis structure cannot specifically expresses the bonding. It describes how several equivalent Lewis structures contribute to one overall hybrid molecule. Resonance hybrids cannot be made to have lower energy than the actual molecules.ĥ. Resonance is a way of describing bonding in chemistry. When separating charge (giving rise to ions), usually structures have little contribution where negative charges are on less electronegative elements, but this may not be true if additional bonds are gained.Ĥ. The Total number of electrons and total charge must be constant.ģ. Position of nuclei must be the same in all structures otherwise they will form isomers.Ģ. If the actual energy of the molecule is E 0 which is determined experimentally then:ġ. From these three energies, E 3 is having lowest energy which corresponds to the most stable resonating structure. The resonance energy is defined as the difference between the electronic energy of a real (conjugated) molecule and a hypothetical Kekul structure with. Let us suppose that a molecule have three resonating structures which have energy E 1, E 2, E 3 associated with them. The meaning of RESONANCE HYBRID is a compound, molecule, ion, or radical exhibiting resonance and having a structure represented in the written form as the average of two or more structural formulas separated each from the next by a double-headed arrow. 6 out of 18 electrons participate in chemical bonds, and the remaining 12 remain as lone pairs. Each oxygen atom has 6 valence electrons, making it a total of 18 for the molecule. The difference between the actual energy of the molecule and that of most stable resonating structure is known as Resonance Energy. Ozone has two major resonance structures that contribute equally to its overall hybrid structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
